Ozempic Face? Try Ozempic Pancreatitis: Inside the GLP-1 Profit Model
Another Fen-Phen? History may not repeat itself, but it sure knows how to rhyme.
The following information is based on a report originally published by A Midwestern Doctor. Key details have been streamlined and editorialized for clarity and impact. Read the original report here.
If you thought “Ozempic face” was bad, wait till you hear what it’s doing inside the body.
A massive study involving 16 million people found GLP-1 users had a 9.09 times greater risk of pancreatitis, 4.22 times greater risk of bowel obstruction, and a 3.67 times greater risk of stomach paralysis.
And if you’ve ever had pancreatitis, it is “quite a painful experience.”
What you’re hearing on the news about Ozempic is still too little, too late.
Here’s the story you’re not getting about Ozempic, the business model behind it, and why a growing number of researchers believe another pharmaceutical disaster is already unfolding in real time.
In early 2023, JP Morgan hosted its annual healthcare conference—a private, invitation-only event it describes as “the industry’s biggest gathering.”
The keynote speakers included the chairman of JPMorgan Chase, the CEO of Eli Lilly, and several managing directors of major healthcare venture capital firms.
The fourth keynote was Dr. Robert Califf.
His day job at the time: Commissioner of Food and Drugs for the United States Food and Drug Administration.
Hmm…
This wasn’t a public health symposium. It wasn’t an academic conference.
It was specifically designed for large investors, and its explicit purpose was to set the pharmaceutical industry’s financial priorities for the year ahead.
A pharmaceutical safety advocate named Kim Witczak obtained what she could from the conference’s public-facing website.
But what was being said behind closed doors?
The conference made two predictions.
Anti-obesity drugs and Alzheimer’s drugs would be the most profitable investment opportunities in the years ahead.
To underscore the point, JP Morgan published a projection showing GLP-1 drug sales—the category that includes Ozempic—growing from $5.1 billion in 2020 to a projected $50.9 billion by 2030.
Then they produced a promotional video to match the energy.
Curiously, Chase deleted it once the original article exposing the conference went public.
This information comes from the work of medical researcher A Midwestern Doctor. For all the sources and details, read the full report below.
The Great Ozempic Scam and The Safe Ways to Lose Weight

Three days before the conference opened, the FDA granted accelerated approval to a controversial new Alzheimer’s drug.
No Advisory Committee meeting. No independent expert panel.
It was approved on a Friday. The FDA Commissioner walked onto the JP Morgan stage on Monday to tell investors the good news about the industry’s prospects.
Once the original article was published exposing Califf’s presence as a keynote speaker, his name was quietly removed from the publicly available keynote list.
That alone tells you something.
What happened in the months after that conference wasn’t subtle.
Ozempic was relentlessly marketed across every demographic.
“Trusted authorities” gave full endorsements. The FDA moved quickly to expand the drug’s approved uses. Clinics began hanging large banners outside advertising they offered it—a sight that remind you of what happen during the opioid crisis.
By 2022, there was a nationwide shortage of the drug.
The manufacturer literally could not produce it fast enough.
Three demographic expansions in particular stand out.
First: the African American community. Ozempic’s manufacturer paid the NAACP to become a lobbyist for the drug. The arrangement was designed so that any attempt to limit Ozempic’s access—or question whether it was appropriate—could be framed as perpetuating systematic racism against the Black community.
It’s a template that would be recognizable to anyone who studied how the tobacco industry worked.
Second: children.
On December 22, 2022—less than three weeks before the JP Morgan conference—the FDA approved semaglutide to treat obesity in children 12 and older.
On January 9, 2023—the opening day of the JP Morgan conference—the American Academy of Pediatrics published official guidelines for treating childhood obesity that strongly endorsed giving GLP-1 drugs to children.
The clinical basis for putting children on a drug that dramatically alters digestion: a 68-week study.
Third: the elderly.
The Medicare Modernization Act of 2003—passed in the wake of the fen-phen disaster—specifically prohibited Medicare from covering drugs prescribed for weight loss.
An “obesity rights” coalition, primarily funded by Ozempic’s manufacturer, has successfully moved a bill through committee to overturn that prohibition.
If passed, it would cost Medicare between $3.1 and $6 billion annually.
The same industry that produced the problem is now lobbying to have taxpayers fund the solution.
The deeper story here isn’t just corruption. It’s a pattern that has played out almost identically before—with a drug called fen-phen—and ended in one of the largest pharmaceutical settlements in American history.
All the details are in the full article from A Midwestern Doctor.
The Great Ozempic Scam and The Safe Ways to Lose Weight

In 1992, a professor who had recently become a director at an FDA division published a study combining two old, marginal weight loss drugs: phentermine and fenfluramine.
His trial ran four years, involved 121 mostly obese women, and showed the combination suppressed hunger and caused weight loss.
He noted that when the drugs were stopped, the weight returned and hunger came back.
He spent almost a decade trying to get it published. A journal finally accepted it in 1992.
What happened next was almost instantaneous.
Word spread like wildfire.
Doctors converted their practices into weight loss pill mills almost overnight. A New York Times article from that era described an infectious disease doctor who paid a chiropractor to hand out blank prescriptions to patients.
Strip mall clinics opened across the country dispensing the drug to anyone who walked in.
A Sacramento psychiatrist devoted his entire practice to fen-phen, buying the drugs wholesale and dispensing them to thousands of patients out of his office.
“A lot of doctors viewed this as a cash register,” he said.
A few months after another version of the drug was rushed to approval in April 1996, reports of heart valve damage began emerging.
When investigators finally assessed the damage, they revealed:
One-third of fen-phen users had already developed asymptomatic heart valve damage—at a point when the drug had only been on the market a few years. Under normal circumstances, similar damage affects roughly 1% of the population.
Over 100 cases of severe heart valve disease were identified in the FDA’s initial search.
The risk of pulmonary hypertension—a serious disease—had increased by up to 30 times. The longer someone had been on the drug, the higher the risk.
The FDA banned it. Over $13 billion in settlements followed.
And the FDA official who ran the original pivotal trial that declared the drug safe later said: “I figured, gee whiz, these drugs have been on the market for 10, 12 years. Everything must be known about them.”
Moving forward, Medicare was banned from covering weight loss drugs.
But the same restriction is currently being lobbied out of existence.
Here’s the aspect of Ozempic that gets the least attention in proportion to how important it is:
Like fen-phen, the weight lost on Ozempic returns once the drug is stopped—and the pivotal clinical trials make this clear if you read them carefully.
Graphs from the drug’s own trials show the weight climbing back the moment participants stopped. The weight regained was proportional to the weight initially lost. A related drug, tirzepatide (Mounjaro), showed the same pattern.
The drug costs $1,000–$1,500 per month.
But it costs less than $5 to manufacture.
And the weight comes back when you stop.
These three facts together constitute the business model.
There’s a data table in A Midwestern Doctor’s article drawn from 16 million patients’ medical records that shows what Ozempic actually does to the body at scale. It deserves more attention than it’s gotten.
Don’t miss it.
The Great Ozempic Scam and The Safe Ways to Lose Weight

Ozempic was engineered to resist breaking down in the body so it only needs to be injected once a week.
The natural GLP-1 protein it mimics has a half-life of 1.5 to 5 minutes. The drug version: approximately 7 days.
Since GLP-1 is responsible for slowing digestion, Ozempic significantly slows the entire gastrointestinal tract for a full week—every week.
A study of 25,617 real-world patients found this causes a 3.5 times increase in the rate of intestinal obstruction.
That’s one study. The larger picture is worse.
The most comprehensive safety data available on GLP-1 drugs was drawn from the medical records of a whopping 16 million patients.
Compared to another weight loss drug not associated with these effects, GLP-1 users showed:
9.09 times greater risk of pancreatitis.
4.22 times greater risk of bowel obstruction.
3.67 times greater risk of gastroparesis—a condition where the stomach becomes permanently unable to empty itself properly. (In many cases after Ozempic, this ends up being permanent.)
1.48 times greater risk of gallstones.
This isn’t a fringe signal from a small study. This is 16 million patients.
Those numbers come from independent research.
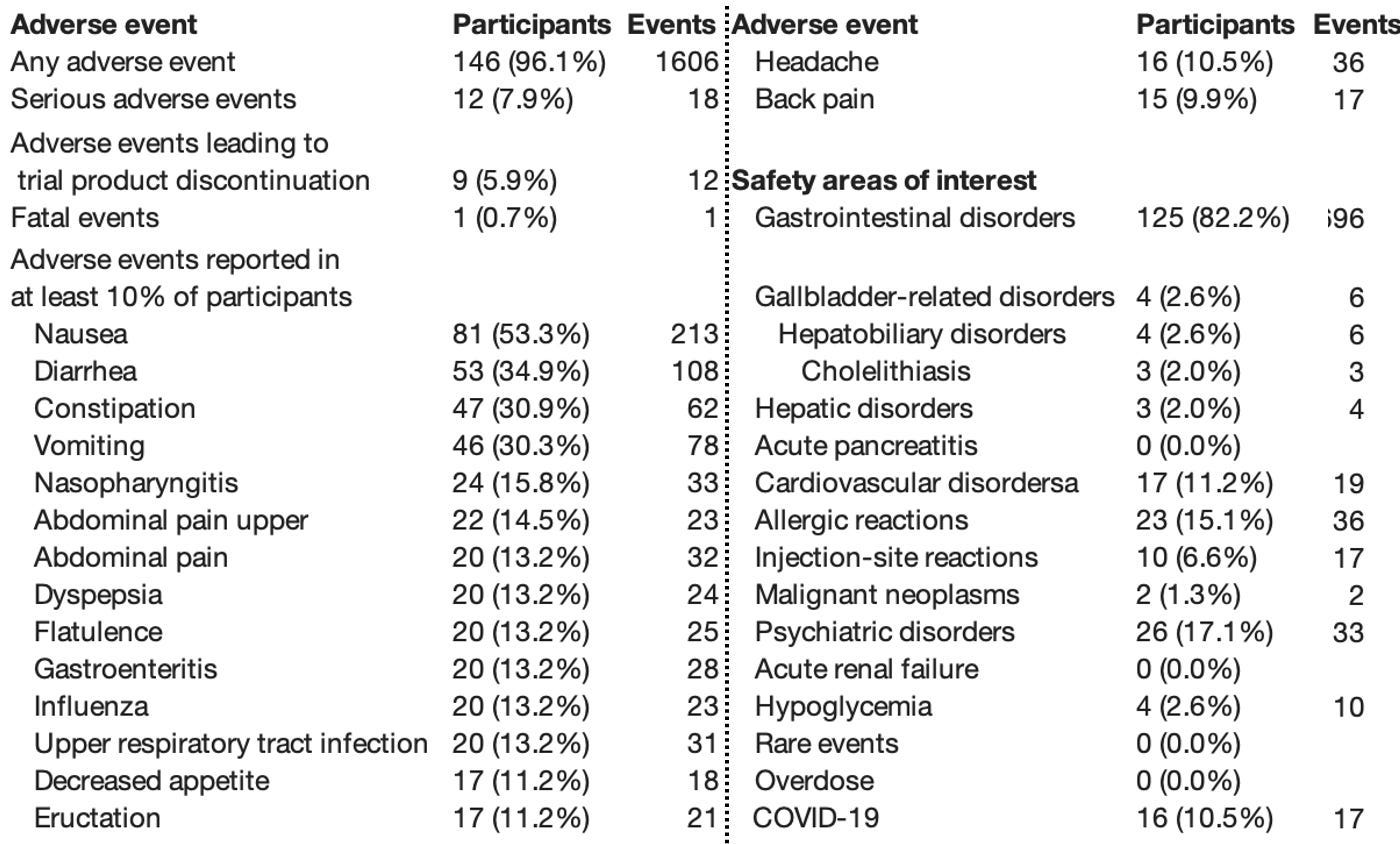
Here is what Ozempic’s own manufacturer reported in their own sponsored trial:
96.1% of participants reported at least one adverse event.
82.2% reported gastrointestinal disorders.
7.9% experienced serious adverse events.
One participant died.
These are the numbers the company chose to publish in support of their drug.
The side effects extend beyond the digestive system.
Lawsuits have been filed over vision loss. Animal studies show Ozempic distorts the architecture of the small intestine. The drug’s own label lists possible thyroid cancer.
And evidence is emerging of a 45% increased risk of suicidal ideation in Ozempic users.
In patients already taking SSRI antidepressants, that number rises to a staggering 345%.
The likely mechanism: Ozempic slows stomach emptying, which alters how psychiatric medications are absorbed—destabilizing patients who are highly sensitive to even small dose changes.
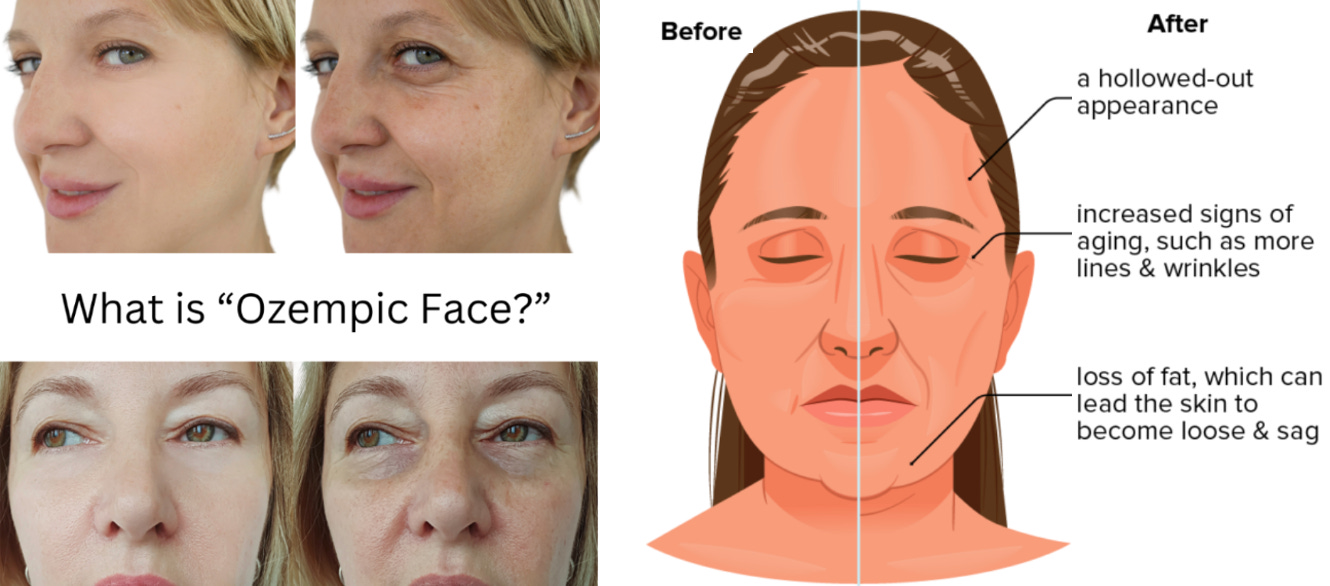
The malnourishment is visible.
Because Ozempic works primarily by suppressing the desire to eat, the body loses fat in a way that is characteristic and recognizable.
The hollowed-out appearance it creates has its own clinical term now.
Ozempic Face.
People are taking this drug to lose weight and “get healthy.” Does Ozempic face look even remotely healthy?
The second half of this article—available to subscribers of A Midwestern Doctor—covers what’s actually driving the obesity epidemic, the causes almost never discussed publicly, and what actually works to reverse it without a lifetime of injections.
It’s worth the read.
The Great Ozempic Scam and The Safe Ways to Lose Weight

Here is what gets lost in all of this:
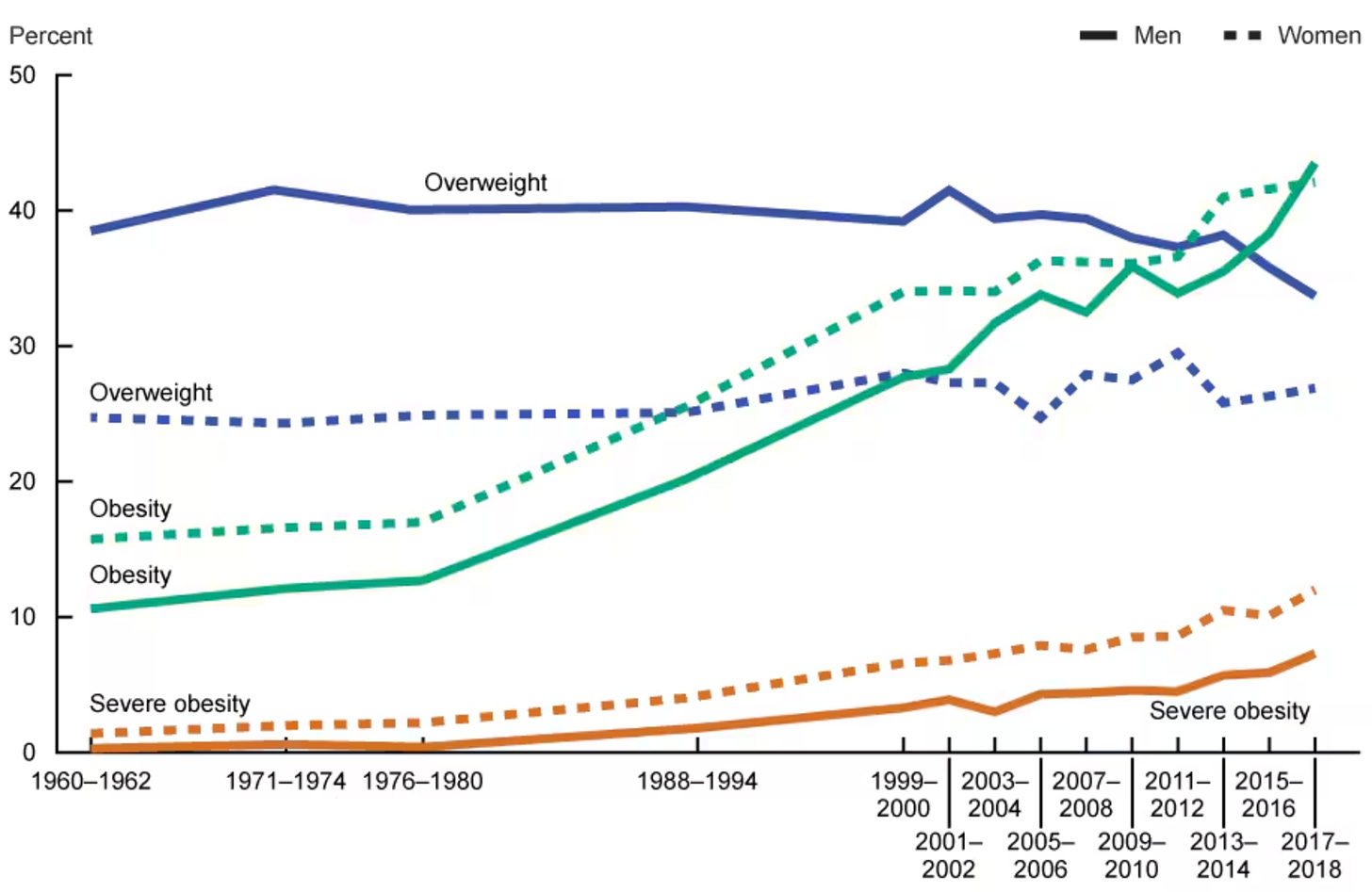
Obesity has been continuously rising for decades. And the official explanation—eat less, move more—has never reversed the trend.
The actual drivers are rarely discussed:
Engineered food additives like seed oils and high fructose corn syrup; estrogen-mimicking compounds from plastics and birth control residue in the water supply; gut microbiome disruption; widespread metabolic and mitochondrial dysfunction; in some cases, immune-mediated damage to insulin production.
All of these causes vary by individual. All of them require actual investigation.
None of them require a lifetime of Ozempic.
Full stop.
The pharmaceutical industry’s ideal product has always been one that provides temporary symptom relief, produces dependence when stopped, and requires perpetual use at a high profit margin.
Ozempic checks every box.
The weight returns when the drug is stopped. The drug costs less than $5 to make and sells for $1,000–$1,500 per month. The side effects—particularly slowed absorption of other medications—create new complications requiring additional pharmaceutical management.
Fen-phen was eventually pulled from the market when the cardiac damage became impossible to ignore.
The question being asked by researchers and physicians paying close attention is: what is the Ozempic equivalent of the heart valve?
It turns out, the pharmaceutical industry doesn’t fear being caught. It fears markets drying up.
Fen-phen killed its users’ heart valves. Thirteen billion dollars in settlements were paid. The principals moved on to the next product. The FDA commissioner who rubber-stamped it was never held accountable. The victims’ stories were quietly absorbed into the legal and financial system—and forgotten.
Now we have a new drug.
Same promise: effortless weight loss.
Same architecture: temporary benefit, permanent customer, severe long-term risk appearing after the market has already been captured.
Same institutional structure: a regulatory agency whose commissioner is on stage at investor conferences previewing the product roadmap.
The only thing different this time is that we have the previous story to learn from.
Whether that actually changes anything is the real question.
Thanks for reading! This information was based on a report originally published by A Midwestern Doctor. Key details were streamlined and editorialized for clarity and impact. Read the original report here.
The Great Ozempic Scam and The Safe Ways to Lose Weight

For a deeper dive into what modern medicine has overlooked—or intentionally buried—check out these other eye-opening reports by A Midwestern Doctor:
The Truth About SSRI Antidepressants
The Hidden Dangers of Hospital Births & How to Protect Your Family
What’s The Healthiest Water To Drink?
While you’re at it, give A Midwestern Doctor a follow. No one brings more research, clinical insight, or historical context when it comes to exposing the health myths we’ve all been fed. This is easily one of the most valuable accounts you’ll ever follow.
If you haven’t subscribed to this Substack yet, take a moment to read what some of the most powerful voices in the medical freedom/truth movement have to say:
“The Vigilant Fox has been putting in a lot of work to create a news platform that shares the stories we want to hear about and brings attention to the most important things to know about. If you want a daily newsfeed in alignment with our ...”
– A Midwestern Doctor, The Forgotten Side of Medicine
“The Vigilant Fox absolutely is on top of things. We must support our fighters, and the Fox is fighting with truth.”
– Tom Renz, Tom Renz’s Newsletter
“Excellent capture of key video presentations on evolving pandemic science.”
– Peter A. McCullough, MD, MPH, FOCAL POINTS (Courageous Discourse)






















Endless cycle ….the cosmetic injector healthcare providers love the cash business that has lined their pockets via GLP-1 side effects …..Endless cycle …. Always follow the money 💰.
Makes me wonder what the former FDA Commissioner Dr. Robert Califf has been up to since he left the FDA. What his bank account and stock portfolios look like......