‘Forever Flu’ Machine: Congress Greenlights Permanent Pandemic Bureaucracy
They’re making sure this becomes the new normal.
This article originally appeared on Jon Fleetwood’s Substack and was republished with permission.
Guest post by Jon Fleetwood
12-week jab machine, nationwide testing grid, and federal “vaccine confidence” propaganda system—backed by permanent funding with no sunset clause.
Congress is advancing legislation that commits at least $19.4 billion over the next five years—while establishing a permanent, multi-billion-dollar annual funding stream with no defined end date—to build a standing, nationwide influenza response system spanning vaccine development, stockpiling, testing, and public behavior campaigns.
U.S. legislators and the Trump administration have already allocated $5.5 billion for a future influenza pandemic through their minibus bill, bringing the government’s recent total committed funding for influenza pandemic orchestration to approximately $24.9 billion across just these two pieces of legislation.
That’s more than the entire gross domestic product (GDP) of Jamaica.
The move comes after President Trump selected a military influenza pandemic policy architect—Dr. Erica Schwartz, who built and enforced surveillance, vaccination, and compliance systems—to take control of the CDC.
The government is orchestrating a multi-billion-dollar influenza pandemic response while simultaneously funding the creation of chimeric influenza pathogens in gain-of-function experiments—the same problem/solution playbook used before the COVID-19 pandemic.
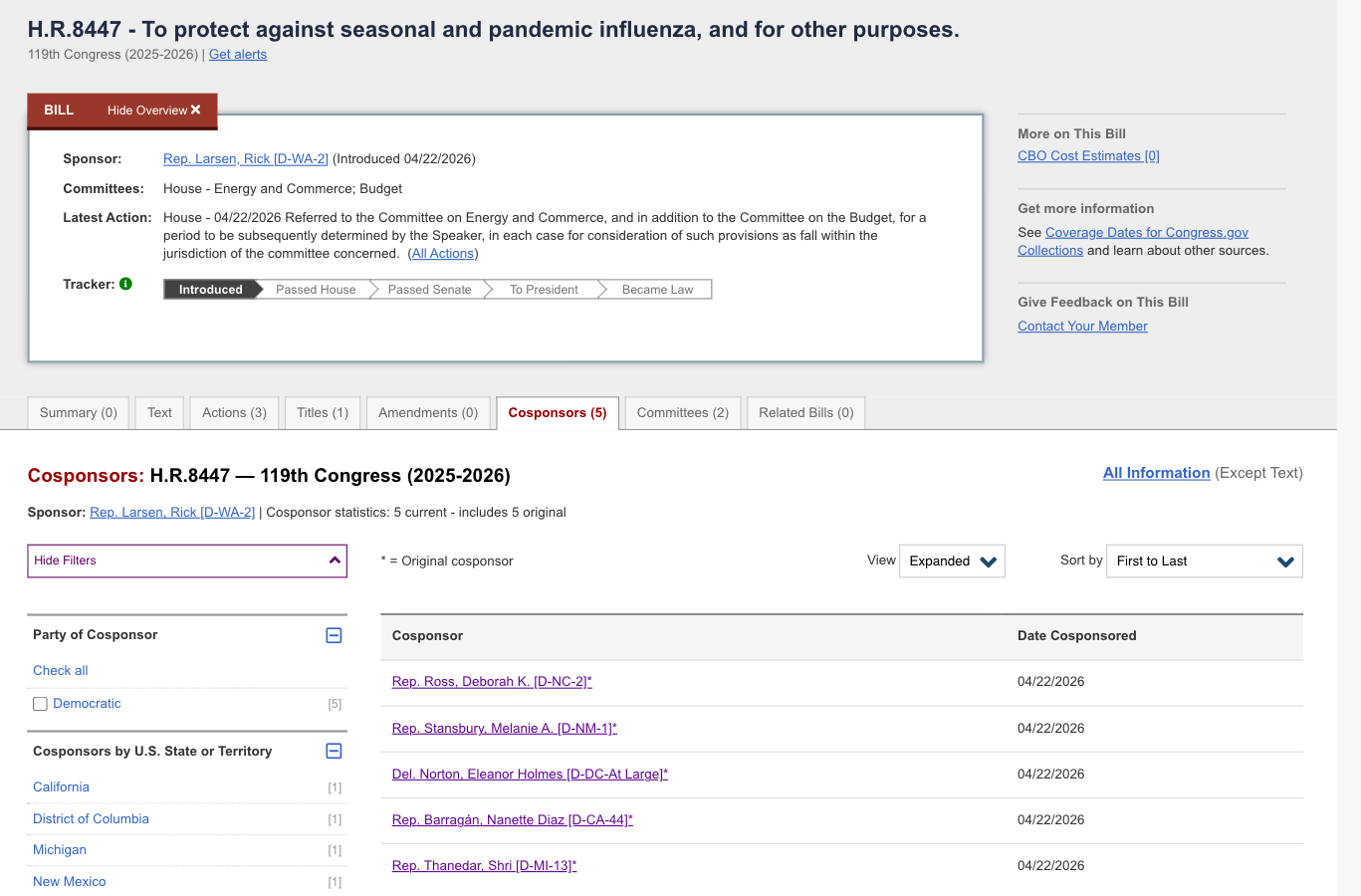
The new bill, titled the “Protecting America from Seasonal and Pandemic Influenza Act of 2026” (H.R. 8447), was introduced the day after Moderna, Inc. announced that it began injecting 4,000 U.S. and UK adults with the company’s investigational mRNA-based H5 pandemic influenza vaccine candidate, mRNA-1018, as part of a Phase 3 study.
Moderna had also just announced that another of its influenza jabs, mCOMBRIAX® (mRNA-1083), had been granted marketing authorization by the European Commission (EC).
H.R. 8447 was introduced in the House by U.S. Representative Rick Larsen (D-WA), along with several co-sponsors, and referred to committee for consideration.
In his press release, Rep. Larsen said he introduced the bill “to prepare for future outbreaks” and “to make sure flu vaccines are available.”
Representatives Deborah Ross (D-NC), Melanie Stansbury (D-NM), Eleanor Holmes Norton (D-DC), Shri Thanedar (D-MI), and Nanette Barragán (D-CA) have signed on as original cosponsors of the legislation.
You can contact these representatives by following the links in their names above.
You can find and contact your representative here.
Critics of the bill warn that it incentivizes future pandemic declarations by locking in a permanent $19.4 billion federal influenza-industrial complex that centralizes rapid vaccine production, testing, stockpiling, and messaging.
They argue it undermines informed consent and individual medical choice by funding aggressive federal campaigns to
boost “vaccine confidence,”
combat “misinformation and disinformation,”
target specific populations with behavioral messaging,
expand vaccine delivery into pharmacies and everyday settings,
link testing directly to immediate treatment pathways,
and frame vaccine uptake itself as a matter of national security—all inside a self-perpetuating system with no sunset clause and billions in guaranteed annual funding.
$19.4 Billion—And That’s Just the Opening Phase
The financial structure is the foundation of the bill.
Limited-Window Funding (2027–2031)
$335 million per year for pandemic influenza countermeasures
→ $1.675 billion over five years
Permanent Annual Funding (No End Date)
$231,358,000—Influenza planning (CDC)
$1,000,000,000—Strategic National Stockpile
$100,000,000—Supply chain and industrial base
$307,000,000—Hospital preparedness
$270,000,000—Universal flu vaccine research
$681,933,000—Immunization programs
$735,000,000—Public health emergency preparedness
$185,000,000—Data modernization
$43,000,000—Advanced molecular detection
→ $3.553 billion per year
Combined Total
$3.888 billion per year (2027–2031)
$19.441 billion over five years
Because most of these authorizations apply to “fiscal year 2027 and each subsequent fiscal year,” the $3.55 billion baseline becomes a permanent fixture unless repealed.
12-Week Vaccine Pipeline Becomes Federal Policy
The bill sets a national requirement that the United States develop the capacity to:
Deliver finished pandemic influenza vaccines within 12 weeks of a new strain emerging
Federal agencies must publish a plan within six months, formalizing a rapid-response vaccine pipeline as standing infrastructure.
A Permanent Push for a Universal Flu Shot
The legislation establishes a national goal to develop a:
“Universal influenza vaccine” within 10 years
The program must be executed in coordination with pharmaceutical manufacturers and supported by ongoing federal investment.
Full Integration of Government & Vaccine Production
HHS is directed to coordinate public-private partnerships across the entire vaccine pipeline:
Ingredients
Manufacturing
Fill-finish
Distribution
Supplies such as syringes and needles
This extends federal coordination into every stage of production and delivery.
Permanent Pandemic Countermeasures Engine
The bill requires the creation of a dedicated federal program to:
Develop vaccines, antivirals, and antibody treatments
Build stockpiles of vaccines, testing supplies, and materials
Expand diagnostic technologies, including at-home testing
Conduct regular pandemic response drills
The system is designed to operate continuously, not just during emergencies.
Centralized Stockpile & Distribution Control
The Strategic National Stockpile is expanded and operationalized to:
Maintain and rotate inventory
Monitor supply levels nationwide
Coordinate distribution during shortages
This places federal authorities at the center of deployment decisions during high-demand periods.
National Testing Grid & Test-to-Treat Model
The bill mandates:
Expansion of at-home and point-of-care testing
A federal plan to rapidly scale diagnostic capacity nationwide
It also requires a test-to-treat demonstration program in so-called high-risk populations, linking testing directly to treatment pathways.
Expansion of Pharmacy-Based Access Points
Federal grants will support state efforts to allow pharmacy personnel to administer:
Vaccines
Tests
Therapeutics
The measure increases the number of distribution and administration sites nationwide.
Ongoing Federal Messaging & Behavior Campaigns
The bill requires:
Annual CDC reporting on strategies to “increase public confidence in the safety and effectiveness of vaccines”
Federal partnerships to address “misinformation and disinformation”
Behavioral research into vaccine hesitancy
Targeted outreach campaigns across digital platforms and communities
The legislation frames vaccine uptake as a priority tied to national outcomes.
Expanded Surveillance & Data Systems
The bill funds:
Public Health Data Modernization Initiative
Advanced Molecular Detection Program
and requires ongoing monitoring of antiviral supply availability across regions.
New ‘Health Defense’ Budget Structure
The legislation creates a “health defense operations” funding designation, allowing agencies like CDC, NIH, and ASPR to:
Submit budget requests directly to the President and Congress
Operate within a dedicated funding classification
Bottom Line
The bill commits $19.4 billion over five years—while locking in a permanent $3.5+ billion annual funding baseline—to build a continuous federal influenza response system that integrates:
Rapid vaccine development and deployment
Long-term universal vaccine research
Centralized stockpiling and distribution
Nationwide testing and treatment infrastructure
Expanded administration through pharmacies
Ongoing public messaging campaigns
Large-scale data and surveillance systems
With most funding authorized indefinitely, the legislation establishes a system designed to operate continuously, expand over time, and remain in place well beyond the initial five-year window.
Copyright 2026 Jon Fleetwood